GORE® Protein Capture Devices with Protein A
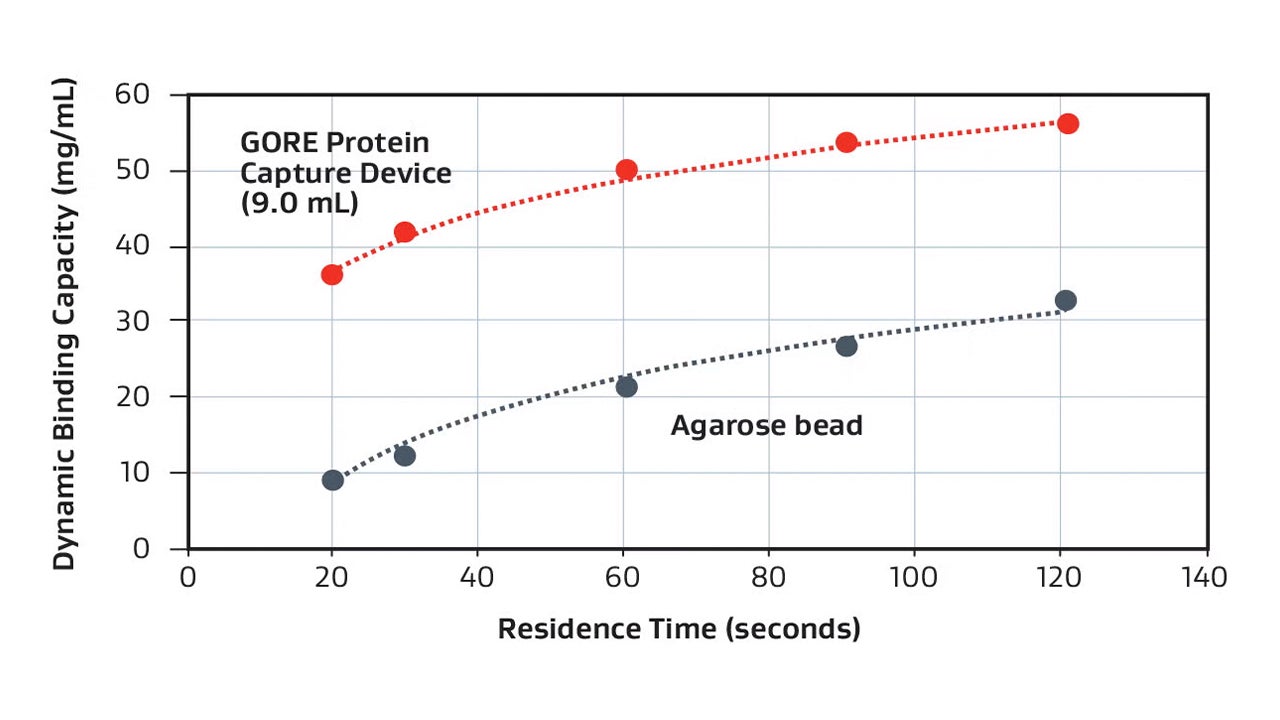
Designed to improve productivity in protein A affinity purification of monoclonal antibodies, GORE Protein Capture Devices provide high binding capacity at short residence time.
Addressing the limitations of traditional Affinity Chromatography
Traditional affinity chromatography technology has not kept pace with increased titers and other upstream process improvements, leading to bottlenecks and lower productivity in downstream protein A purification. To shorten processing times, traditional resin-based protein A is often over-sized or underutilized – simply to keep up.*
*Reference: “Disrupting Downstream Process Bottlenecks.” Genetic Engineering, June 14, 2018.

Learn About Our Protein Membrane Employed in a Single-Use Platform
Technical Specifications
GORE Protein Capture Devices with immobilized Protein A* are designed to enhance downstream purification by improving productivity. These pre-packed columns feature a unique membrane that delivers high dynamic binding capacity with short residence times. Designed to increase the efficiency of protein A column purification processes, the Devices accommodate rapid cycle capability at low pressure drop, along with demonstrated ability to withstand multiple cleaning cycles. This increased productivity offers researchers the potential for a faster path to clinical trials.
| Binding Capacity and Cycling Information | ||
|---|---|---|
| Dynamic binding capacity | ≥ 30 mg/mL | ≥ 40 mg/mL |
| Residence Time | 20 seconds | 30 seconds |
| Validated Cleaning Cycle Details | 0.2 N NaOH for 3 minutes up to 100 cycles | 0.2 N NaOH for 3 minutes up to 100 cycles |
*recombinant Protein A
The GORE Protein Capture Device scales across sizes without compromising binding capacity or speed to quickly and effectively capture antibodies as shown in this overlay of a 1 mL, 9 mL and 58 mL bind-elute curve and dynamic binding capacity graph.
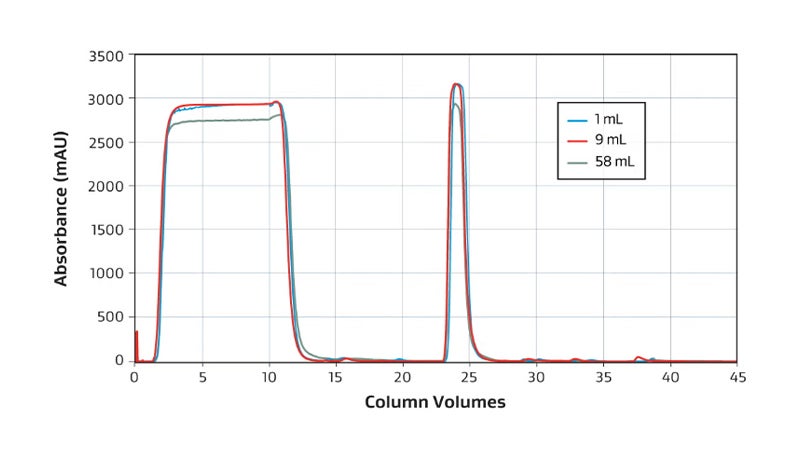
Overlaid chromatogram showing 1 mL (blue), 9 mL (red) and 58 mL (green) UV absorbance.
The difference in UV of harvest reflects different LC instruments.
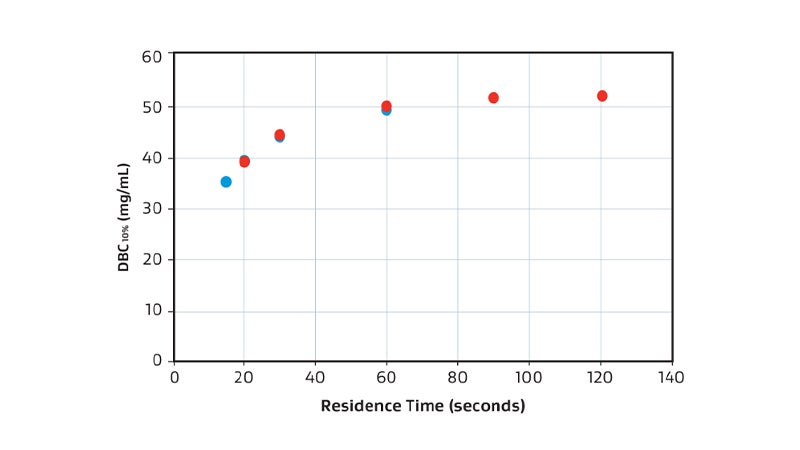
Overlaid DBC10% comparison of 58 mL (red) and 250 mL (blue).
The GORE Protein Capture Devices with Protein A can streamline antibody purification processes because they incorporate a proprietary membrane composite. Unlike traditional support matrices, the composite membrane bed maintains stable performance across a wide range of flow rates, avoiding collapse, channeling, or structural changes.
Pressure Drop
for a 58 mL Device (PROA201)
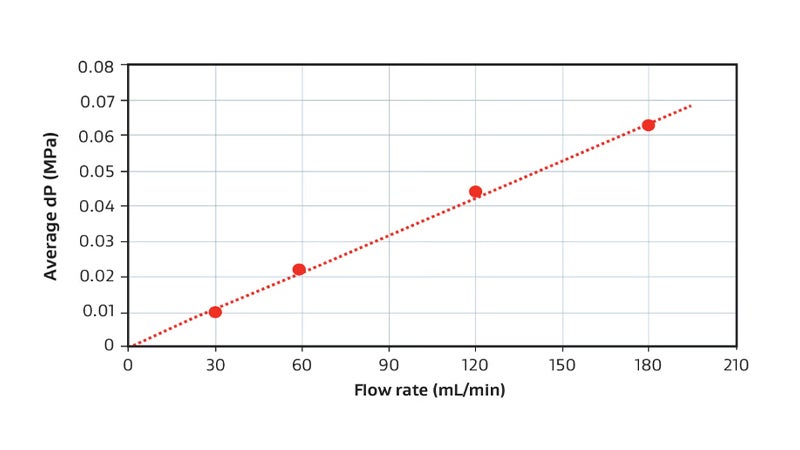
Example pressure drop vs. flow rate relationship for a GORE Device when using phosphate buffer at room temperature. For reference, 116 mL/min equates to 30 seconds residence time.
Pressure Drop
Consistency
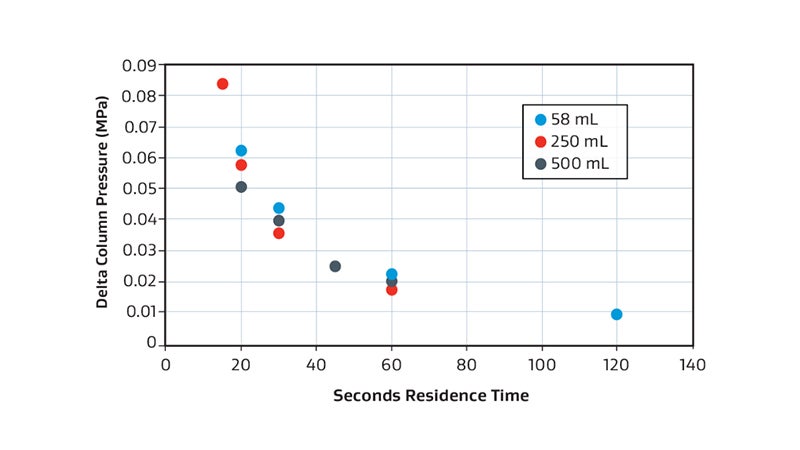
Example of pressure drop vs. residence time consistency for the 58 mL (blue), 250 mL (red) and 500 mL (grey) when using phosphate buffer at room temperature.
GORE Protein Capture Devices are manufactured following the applicable material quality and regulatory requirements, including relevant Good Manufacturing Processes as defined in the Gore PharmBio quality system which is certified to ISO 13485 and ISO 15378. For current applicable compliance statements and quality control information contact us.
| Gore Part Number | |||
|---|---|---|---|
| Components | PROA101 PROA102 PROA103 | PROA201 PROA301 | PROA202 PROA302 PROA303 |
| Device housing components | Polypropylene | Polypropylene and Polyether ether ketone (PEEK) primarily* | |
| Frit material(s) | Polypropylene | Polyether ether ketone (PEEK) | |
| Membrane support matrix | Proprietary composite | ||
| Ligand | Recombinant Protein A | ||
| Connectors | 10-32 threaded fittings (PROA101/PROA102) | 5/16-24 Flat-bottom fittings (PROA201) | 3/4" Tri clamp (0.984" (OD)) |
| 10-32 coned thread port (PROA103) | 1/2-20 flat-bottom threaded fittings (PROA301) | ||
GORE Protein Capture Devices are compatible with standard chromatography systems, and are available in sizes as listed in table below. Contact Gore for pricing and availability. These Devices for protein A purification are not available in all regions.
Part Number/Ordering Information
| Part Number | PROA101 | PROA102 | PROA103 | PROA201 | PROA202 | PROA301 | PROA302 | PROA303 |
|---|---|---|---|---|---|---|---|---|
| Description | 1.0 mL Device | 3.5 mL Device | 9.0 mL Device | 58 mL Device | 116 mL Device | 250mL Device | 500 mL Device | 1L Device |
See Validation Guides for performance specifications and evaluations
GORE Protein Capture Devices are manufactured following the applicable material quality and regulatory requirements, including relevant Good Manufacturing Processes as defined in the Gore PharmBIO quality system which is certified to ISO 13485 and ISO 15378. For current applicable compliance statements and quality control information contact us.
Explore More
NOT INTENDED FOR USE
In medical device or food contact applications or with radiation sterilization.
Prefer to Call?
Have questions or unique requirements?
Our experts are here to guide you.